|
Finally, IR is a non-destructive technique, meaning the sample is not harmed and can often be recovered afterward. The wide range of suitable sample types also makes infrared spectroscopy incredibly versatile. Analysis can be performed very quickly, leading to high throughput and allowing for several samples to be tested in quick succession. Possibly the greatest advantage of IR spectroscopy is its speed. Advantages and limitations of IR spectroscopy Solid samples can be crushed into fine powder to achieve the best results. If the sample is to be tested in solution, care must be taken to ensure the solvent does not absorb infrared energy in the same region as the sample, as this may mask important information. IR spectroscopy can be performed on solids, liquids, and solutions, provided they are of a high enough purity. This provides key information about the chemical structure of the sample. Portions of the infrared spectrum are absorbed during this process, with each absorbance corresponding to a specific type of bond and movement.īy measuring the infrared light transmitted through the molecule and taking notes of the sections of the spectrum that are absorbed, it is possible to determine which bonds are present. Some might stretch, bend or even rock very rapidly.  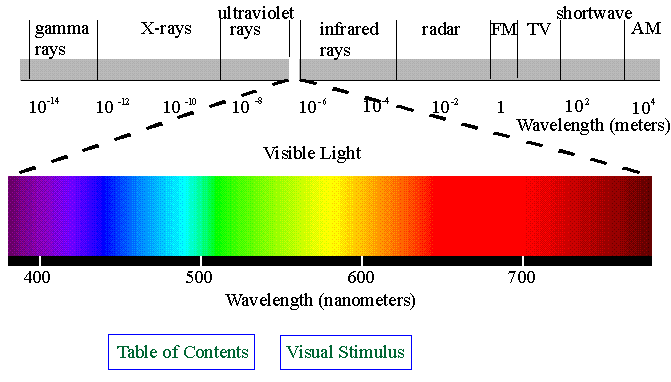
When the beam interacts with different chemical species in the sample, it will provide enough energy to cause the bonds in those molecules to vibrate in a variety of different ways. In its simplest terms, IR spectroscopy works by passing a beam of infrared light through a sample. Infrared spectroscopy is also used across all biochemical industries, ranging from medical to food, to test for selected proteins and other organic compounds. IR spectroscopy is commonly used in pharmaceuticals and other consumer industries to quickly test samples for unwanted contaminants.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
